 /GettyImages-141483984-56a133b65f9b58b7d0bcfdb1.jpg)
Niels Bohr and the Quantum Atom: The Bohr Model of Atomic Structure 1913-1925 (Oxford University Press, 2012) the Aaserud-Heilbron book cited in Ref. This is an excellent idea, given the historic importance of Bohr. Produced by: Experimentarium in corporation with the Niels Bohr Archive. Many teachers like to introduce the Bohr atom toward the end of an introductory physics course. Petersen from the Niels Bohr Institute and director of the Niels Bohr Archive Finn Aaserud celebrate the 100 years of the atomic model. Mainly because it solved two mysteries, namely: why atoms radiated certain colours of light when heated and why the atom consisting of a positive as well as a negative electrical charge did not self-destruct. But eventually leading physicists of the time became convinced of its value. As a result Niels Bohr’s model of the atom was not generally accepted at first. It was primarily based on the concept of energy levels or shells, where electrons were assumed to orbit the nucleus in specific circular paths. He won the Nobel Prize in Physics in 1922. Niels Bohr was born in Copenhage and best known for his work in atomic theory. The element bohrium is named in his honor. In 1913, Niels Bohr published his atomic model, which revolutionized our understanding of the structure of the atom. The ingenious idea Niels Bohr used in his model was to quantize the atom – a brave idea as the world was considered classical and not quantized. The Bohr Atomic Model, proposed by Niels Bohr in 1913, was a significant advancement in understanding atomic structure. Bohr and his wife Margrethe had six children, one of whom, Aage Niels Bohr, became a very successful physicist and also won a Nobel Prize in Physics. The model was the first consistent description of how electrons contributed to constitute matter. 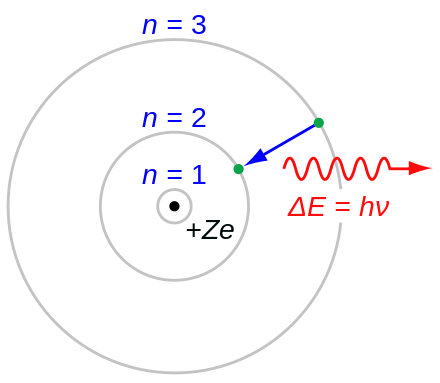
He realized early that the research front in theoretical physics was moving from the study of the atom as a whole to the study of its nucleus. liquid-drop model, in nuclear physics, a description of atomic nuclei in which the nucleons (neutrons and protons) behave like the molecules in a drop of liquid. Niels Bohr’s model of the atom revolutionized physics when it was published in 1913. In the early 1930s Bohr found use once more for his fund-raising abilities and his vision of a fruitful combination of theory and experiment. 100 years anniversary for Niels Bohr's Atomic Model 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
